
So I got the question marked incorrect which probably means I didn't do the calculation for copper's bond strength correctly. Simply put, a hydrogen bond is an attraction between a slightly positive hydrogen on one molecule and a slightly negative. Ionic > covalent > metallic > H-H (van der Waals) Hydrogen bonds are the strongest of all intermolecular forces. polar solvent could help to alter the macroion-counterion interaction, and as a result. There are of course numerous exceptions on both ends. D2O is more structured with stronger hydrogen bonds than. Ionic bonds result from the mutual attraction between oppositely charged ions while a Covalent Bond is a bond that results from a sharing of electrons between nuclei. A quick survey finds that most covalent bonds are 200-300 kJ/mol and most lattice energies are >600 kJ/mol. Ionic bonds are stronger than covalent bonds due to following reasons. non-interacting) ions and the ionic solid. There are four types of bonds or interactions: covalent, ionic. 
It is important to understand the various types of bonds because they help determine how different molecules function within an organism. These bonds are interactions between two atoms that hold the atoms together. I should probably also note that based off of copper's heat of vaporization of 3630 J/g and its molar mass of 63.546 g/mol I calculated a bond strength of 383 zJ and WRONGLY concluded: The equivalent for ionic compounds is the lattice energy, which is what I said above: the difference in energy between the gaseous (e.g. Atoms can form several types of chemical bonds. So is it just the case that Na-Cl is a particularly strong ionic bond and H-O is a particularly weak covalent bond such that this particular ionic bond is stronger than this particular covalent bond? Or are the other answers incorrect? To complicate things further, this question has been asked numerous times in various iterations and other answers have stated that covalent bonds are stronger than ionic bonds, which are in turn stronger than metallic bonds. It may be helpful to consider the size of the atoms too, smaller atoms (hydrogen) would have stronger bonds, and. I tried specifically looking for copper, silver, and iron and couldn't find the bond strength between atoms. The hydrogens have a more polar bond, so their electrons are distributed in a way that causes their bond to be stronger, whereas the ion-dipole bond has a greater distance between their electrons so their bond is weaker. What I cannot find is the bond strength for metal-to-metal atoms. I've found the exact bond strength of 3 of 4 of these. The important alkali metal ions Na and K lose their distinctive properties if the hydrogen bonds are 11 stronger or 11 weaker respectively. b) Hydrogen bonds are stronger than covalent bonds. The bond energy of the covalent OH bonds in water is 458.9 kJ/ mol, which is the average of the values. dipole-dipole, London Dispersion, ionic, and hydrogen-bonding. For example, an HOH bond of a water molecule (HOH) has 493.4 kJ/mol of bond-dissociation energy, and 424.4 kJ/mol is needed to cleave the remaining OH bond. "Rank the following bonds from strongest to weakest and provide the bond energy: the bond between hydrogen an oxygen in a water molecule the bond between sodium and chloride in the NaCl molecule the bond between atoms in a metal the van der Waals bond between adjacent hydrogen atoms." The bond between ions of opposite charge is strongest when the ions are small. This is problem 4.10 from the book "Nanotechnology: Understanding Small Systems" 2nd ed. Not looking for someone to do my homework for me, just trying to flesh out an area where I'm not yet proficient. 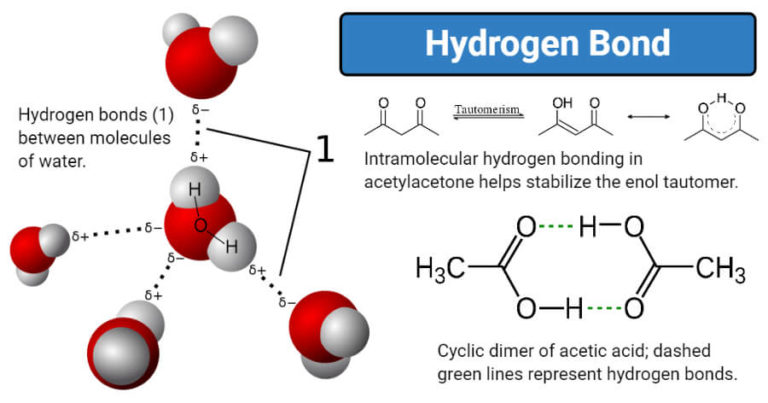
(CC BY-SA-NC anonymous) The green arrows pointing towards each other represent attraction.Note: I've already handed this in for homework and got the question wrong but don't understand why. \): Both attractive and repulsive dipole–dipole interactions occur in a liquid sample with many molecules.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
